E170II
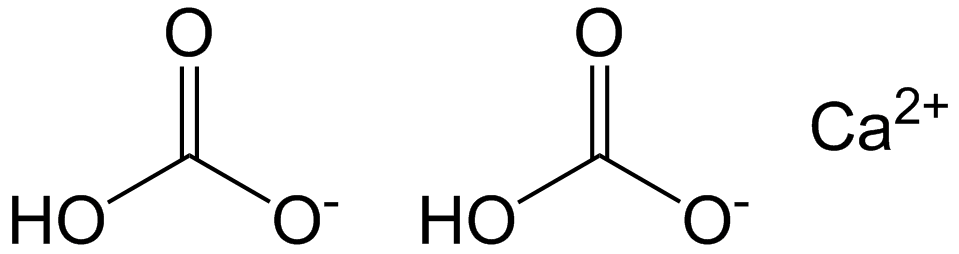
E170ii - Calcium hydrogen carbonate
✅ Risk Level: Safe • Additive
About this additive
Origin
Calcium hydrogen carbonate, also known as calcium bicarbonate, is an inorganic salt formed by the reaction of calcium carbonate with carbon dioxide and water. It exists only in solution and cannot be isolated as a solid compound. It is commonly found in natural water sources, particularly in regions with limestone or chalk deposits. The formation of calcium hydrogen carbonate is crucial in the natural carbon cycle and contributes to the hardness of water.Usage
In the food industry, calcium hydrogen carbonate is not directly used as an additive in the same way as calcium carbonate (E170i). However, it plays an indirect role. It is primarily significant in the brewing industry, where it affects the pH and mineral content of the water used for brewing. The presence of calcium hydrogen carbonate can influence the extraction of flavors from malt and hops, impacting the final taste and stability of beer. It can also be used in the preparation of mineral waters.Side Effects
Since calcium hydrogen carbonate exists primarily in solution and is naturally present in drinking water, it is generally considered safe for consumption at typical levels. High concentrations in drinking water contribute to water hardness, which can lead to scale formation in pipes and appliances but poses no direct health risks. There is no established ADI (Acceptable Daily Intake) for calcium hydrogen carbonate, as it is a naturally occurring compound and not intentionally added to food in significant quantities.Regulatory Status
Calcium hydrogen carbonate is not regulated as a direct food additive in the same manner as calcium carbonate (E170i). Its presence in food and beverages is primarily due to its natural occurrence in water sources. Regulations regarding water quality, including mineral content, may indirectly affect the levels of calcium hydrogen carbonate in drinking water and beverages. Calcium carbonate (E170i), from which calcium hydrogen carbonate is derived in aqueous solutions, is permitted as a food additive in the EU and other regions, subject to specific purity criteria and usage levels.Sources
- 📚 EFSA
- 📚 FDA
- 📚 Wikipedia
- 📚 PubChem
Quick Facts
Type
Additive
Risk Level
✅ Safe
Vegan?
✅ Yes
Halal?
❓ Maybe
